Dual use research refers to scientific activities conducted for legitimate purposes that generate knowledge, information, technologies, and/or products that can be utilized for both benevolent and harmful purposes. Effective oversight of dual use research is based on identifying associated risks and devising ways to mitigate these risks.
The 2024 United States Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential (USG Policy) consolidates and replaces the 2012 Federal DURC Policy, the 2014 Institutional DURC Policy, and the 2017 P3CO Framework, creating a single, streamlined framework for governance. It provides an oversight framework for research with biological agents or toxins that is within what is referred to as Category 1 or Category 2 research.
The UCLA IRE is the body charged with oversight and education of life sciences research that falls within the scope of the USG Policy. The IRE advises and reports to the Chancellor through the Vice Chancellor for Research and Creative Activities.
Although the 2024 USG Policy applies specifically to federal departments and agencies that fund or sponsor intramural or extramural research with biological agents or toxins where the research is within Category 1 or Category 2, it is expected that institutions implement oversight of non-federally funded Category 1 and Category 2 research in accordance with the framework under the USG Policy.

Definitions
Dual Use Research
Dual use research is research conducted for legitimate purposes that generates knowledge, information, technologies, and/or products that can be utilized for benevolent or harmful purposes.
Dual Use Research of Concern (DURC)
DURC is life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be misapplied to do harm with no, or only minor, modification to pose a significant threat with potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, materiel, or national security.
Federal Funding Agency (FFA)
The FFA is a federal department, agency, institute, center, or office that funds or sponsors intramural or extramural research at research institutions in the United States or internationally, with biological agents or toxins where the research is within Category 1 or Category 2 under the USG Policy.
Institutional Contact for Dual Use Research (ICDUR)
The ICDUR is the official designated by the research institution to serve as an internal resource for application of this Policy as well as the liaison (as necessary) between the institution and the relevant federal funding agency.
Institutional Review Entity (IRE)
The IRE is the entity established by the research institution to execute the institutional oversight responsibilities described in the USG policy.
Pathogen with Enhanced Pandemic Potential (PEPP)
PEPP is a type of pathogen with pandemic potential (PPP) resulting from experiments that enhance a pathogen’s transmissibility or virulence, or disrupt the effectiveness of pre-existing immunity, regardless of its progenitor agent, such that it may pose a significant threat to public health, the capacity of health systems to function, or national security. Wild-type pathogens that are circulating in or have been recovered from nature are not PEPPs but may be considered PPPs because of their pandemic potential.
Pathogens with Pandemic Potential (PPP)
PPP is a pathogen that is likely capable of wide and uncontrollable spread in a human population and would likely cause moderate to severe disease and/or mortality in humans.
Risk Mitigation Plan (RMP)
Developed by the IRE in partnership with the PI, the RMP describes measures to be instituted for the conduct and communication of Category 1 and Category 2 research. The RMP will include details of the risks identified by the IRE in its review of the research, and an explanation of the risk mitigation strategy or strategies that are being implemented to address those risks.
Mandate
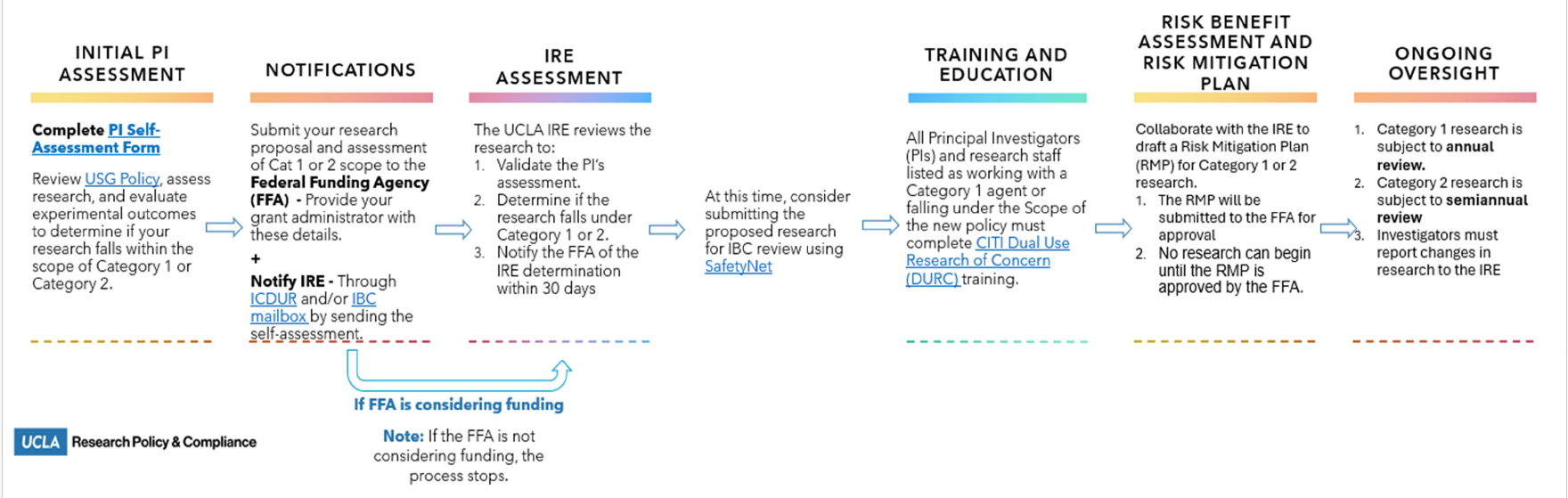
The IRE ensures institutional compliance with the USG Policy through oversight of all UCLA research classified as Category 1 or Category 2.
- Education and Training for PIs: The IRE shall ensure that no research classified as Category 1 or Category 2 is conducted unless PIs have completed required education and training. Training must enable PIs to conduct an initial self-assessment to determine if their research meets the criteria for either category.
- Risk Identification and Assessment: The IRE will work with PIs to assess whether research involving relevant biological agents or toxins produces, aims to produce, or is reasonably anticipated to produce one or more experimental outcome of concern as defined by the USG Policy. For research classified as Category 1 or Category 2, the IRE will determine the level of oversight and necessary risk mitigation strategies.
- Conduct of Risk-Benefit Assessment and Development of Risk Mitigation Plans (RMPs): The IRE will collaborate with the PI to perform a risk-benefit assessment to evaluate the potential benefits and risks associated with proposed Category 1 and Category 2 research. Based on this assessment, the IRE and PI will develop a draft RMP to guide conduct and communication of the research and define strategies to mitigate identified biosafety and biosecurity risks. The draft RMP must be reviewed and approved by the relevant federal funding agency prior to the initiation of research.
- Ongoing Monitoring and Review: The IRE will conduct continuing reviews of all active Risk Mitigation Plans in conjunction with the required progress report submissions, as outlined in the policy. Reviews will occur annually for Category 1 research and semi-annually for Category 2 research to ensure compliance with the USG Policy and adapt plans as necessary to address evolving risks or changes in research scope.
Committee Structure and Administration
IRE Membership
The Institutional Review Entity (IRE) is composed of individuals with the scientific expertise and policy knowledge necessary to assess the dual use and pandemic potential of life sciences research conducted at UCLA.
Membership includes subject matter experts across relevant research disciplines to ensure appropriate scientific breadth, as well as individuals with experience in biosafety, biosecurity, and risk management. The IRE also includes the Institutional Contact for Dual Use Research (ICDUR) and the Biosafety Officer (BSO) as standing members to support coordination across biosafety and compliance functions.
To ensure continued alignment with federal oversight expectations, the IRE will periodically review its membership structure and adjust as needed to maintain appropriate representation based on the evolving scope of research and policy updates.
Committee Administration
Administrative and operational support for the IRE is provided by the Office of Research Policy and Compliance (RPC) under the Office of the Vice Chancellor for Research and Creative Activities.
IRE Meeting Schedule
The IRE convenes full meetings on an as-needed basis to review research that may fall under Category 1 or Category 2, particularly when a PI self-assessment indicates the possibility of experimental outcomes covered by the policy. In addition, the IRE meets at least annually to:
- Review ongoing oversight responsibilities;
- Assess the effectiveness and currency of institutional policies and procedures;
- Ensure that the committee remains aligned with federal policy expectations.
To streamline the review process and respond efficiently to time-sensitive or routine matters, the IRE may also conduct reviews electronically (e.g., via email).